Grade LevelHigh School |
TopicsChemistry |
AuthorsKelsey Lucas, Kristen L. Cacciatore |
Lesson Summary
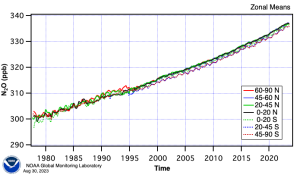 This lesson addresses reaction kinetics and collision theory in the context of research into bacterial denitrification processes under varying conditions. It promotes student understanding of temperature effects on reaction rate and informs students about how kinetics can be applied to understand phenomena related to climate change.
This lesson addresses reaction kinetics and collision theory in the context of research into bacterial denitrification processes under varying conditions. It promotes student understanding of temperature effects on reaction rate and informs students about how kinetics can be applied to understand phenomena related to climate change.
Upon completion of this lesson, students will be able to:
- create and analyze graphs related to rates of a chemical reaction in order to understand temperature effects on the reaction.
- explain how reaction-rate data can be used to study a natural process that could potentially be used to reduce levels of a greenhouse gas.
Standards
Science and Engineering Practices:
SP4MA Science and Technology/Engineering (2016):
HS-PS1-5NGSS (2013):
HS-PS1-5Common Core Math/Language Arts Standards:
CCSS.ELA-LITERACY.RST.9-10.1Tags
aerobic, anaerobic, chemical reactions, collision theory, denitrification, reaction rateEducator SoundBites
Feedback coming soon!
Lesson Documents
Bite
Student
Educator
Feedback
Let us know what you think of this lesson!